Stainless steel corrosion costs industries billions annually. The problem is not that stainless steel corrodes easily. Rather, engineers often fail to recognize that six distinct corrosion mechanisms exist, each with unique causes and prevention methods.
Understanding these differences matters because prevention strategies that work for one corrosion type can be useless, or even counterproductive, for another. This guide covers each corrosion type, including visual identification cues, environmental triggers, grade selection by PREN values, and actionable prevention strategies you can specify directly.
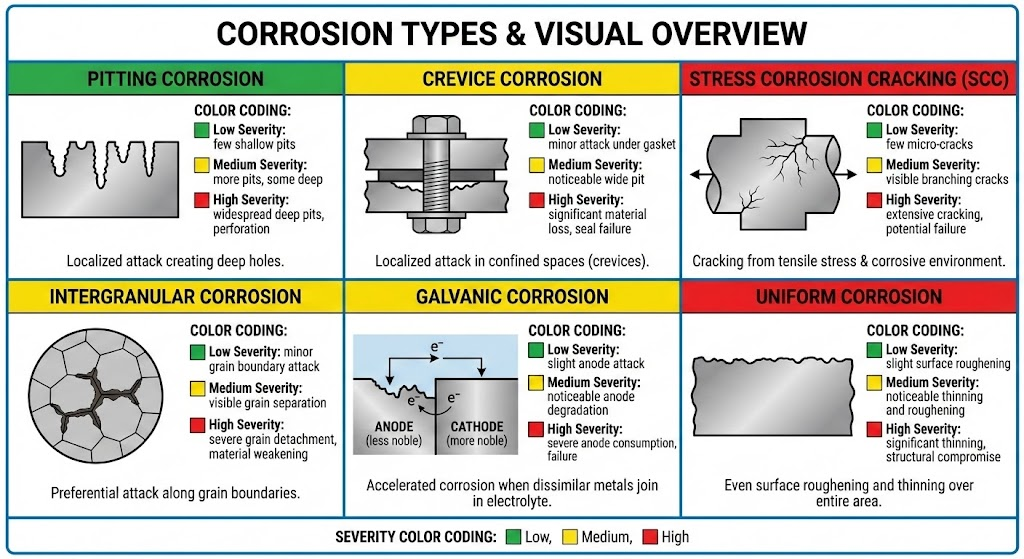
Six main corrosion types affect stainless steel, each triggered by specific environmental conditions. The table below provides a quick reference for identifying which type you may be dealing with.
| Type | Primary Cause | Visual Signature | Severity |
|---|---|---|---|
| Pitting | Chlorides + oxygen | Small, deep holes | High |
| Crevice | Oxygen depletion in gaps | Wide, shallow pits | High |
| Stress Corrosion Cracking (SCC) | Tensile stress + chlorides + heat | Branching cracks | Critical |
| Intergranular | Sensitization at grain boundaries | “Sugary” texture, weld decay | Moderate-High |
| Galvanic | Dissimilar metals in contact | Accelerated attack at junction | Moderate |
| General/Uniform | Strong acid attack | Even surface loss | Variable |
Pitting corrosion creates localized attack from chloride ions. Small diameter cavities form on the surface, often with reddish-brown oxide deposits.
Crevice corrosion occurs when oxygen depletes in tight gaps under gaskets, clamps, or fasteners. The pits are typically wider but shallower than pitting.
Stress corrosion cracking (SCC) requires three simultaneous conditions: tensile stress, chloride ions, and elevated temperature. Above 80C, SCC can initiate in hours rather than months, making even brief temperature excursions dangerous.
Intergranular corrosion attacks chromium-depleted grain boundaries. It commonly appears as “weld decay” in the heat-affected zone (HAZ) adjacent to welds.
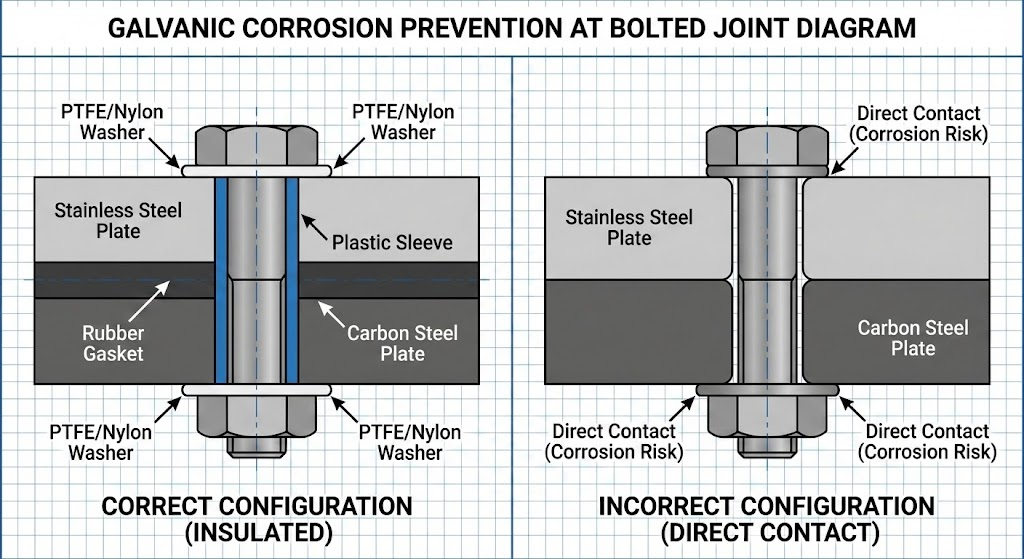
Galvanic corrosion accelerates attack on the less noble metal when two dissimilar metals contact in the presence of an electrolyte.
General corrosion uniformly attacks the entire surface when exposed to aggressive acids, particularly hydrochloric or mid-concentration sulfuric acid.
Visual identification helps engineers determine root causes and select appropriate countermeasures. Each corrosion type leaves distinctive marks.
| Type | Appearance | Location | Key Identifier |
|---|---|---|---|
| Pitting | Small cavities, reddish-brown deposits | Random spots on exposed surfaces | Entry point small but damage extends deep |
| Crevice | Wide, shallow pits | Under gaskets, clamps, fasteners | Only visible when disassembled |
| SCC | Fine branching cracks | Stressed areas, welds, bent sections | Crack pattern with little visible corrosion product |
| Intergranular | Grain boundary etching, “sugary” texture | Heat-affected zones near welds | Parallel to weld line |
| Galvanic | Accelerated attack on less noble metal | Junction between dissimilar metals | Sharp demarcation at contact boundary |
| Uniform | Even surface attack, consistent roughening | Entire exposed surface | No localized pits or cracks |
Pitting creates small diameter cavities that extend deep below the surface. The pit opening often appears smaller than the cavity beneath. Look for reddish-brown oxide deposits around pit mouths on exposed surfaces.
Random distribution across the surface is typical. Pits often initiate at surface defects, inclusions, or areas where the passive layer has been damaged.
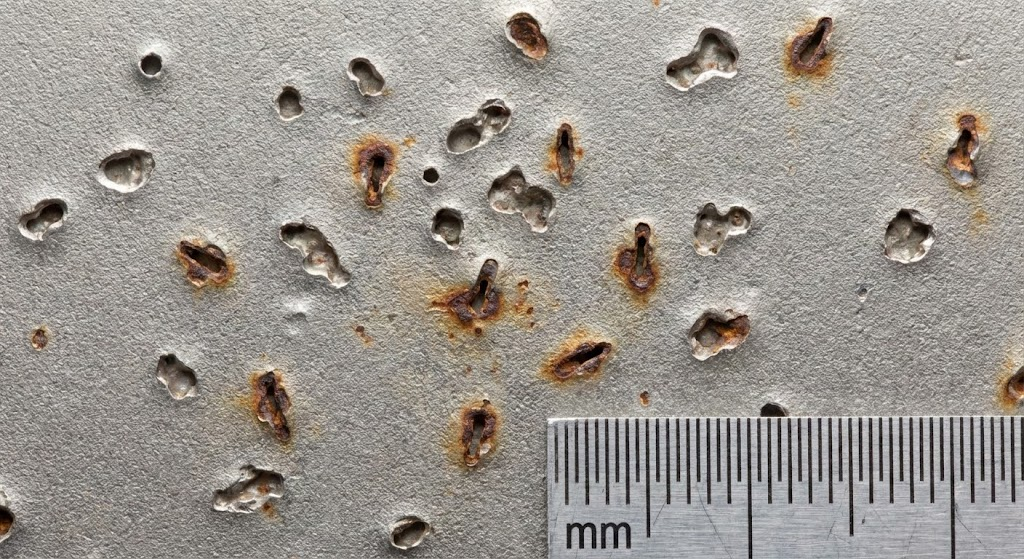
Crevice corrosion produces wider, shallower pits than pitting. The damage follows the geometry of the crevice itself and is often invisible until components are disassembled.
I recommend prioritizing crevice elimination through design over upgrading material grades. The critical crevice corrosion temperature (CCCT) is consistently 15-20C below the critical pitting temperature (CPT) for any given alloy, making geometry more critical than material selection alone.
SCC produces fine, branching cracks oriented perpendicular to the direction of tensile stress. Unlike other corrosion types, SCC often shows minimal visible corrosion products.
Look for cracks in stressed areas, particularly near welds, bent sections, and cold-worked regions. The branching pattern distinguishes SCC from mechanical fatigue cracks, which are typically straighter.

Intergranular attack creates a distinctive “sugary” texture when severe. Grain boundary etching appears first, potentially progressing to grain dropping in extreme cases.
The damage pattern runs parallel to weld lines, concentrated in the heat-affected zone. This is why intergranular corrosion is often called “weld decay.”
Galvanic corrosion shows accelerated attack concentrated on the less noble (anodic) metal. A sharp demarcation line at the metal contact boundary is the distinguishing feature.
The Statue of Liberty provides a famous example: after 100 years, the original shellac insulation between its copper skin and iron framework failed, causing extensive galvanic corrosion. Engineers replaced it with PTFE during restoration.
Uniform corrosion attacks the entire exposed surface evenly. Look for consistent roughening or metal loss across the area without localized pits or cracks.
This corrosion type is actually the easiest to predict and manage because attack rates are relatively uniform and measurable.
Each corrosion type has specific environmental triggers. Understanding these thresholds helps you match materials to operating conditions.
| Type | Temperature Factor | Chloride Role | Other Triggers |
|---|---|---|---|
| Pitting | Higher = faster initiation | Critical – as low as 25 ppm | Low pH, stagnant conditions |
| Crevice | Lower threshold than pitting | Accelerates attack | Oxygen depletion geometry |
| SCC | Above 60C for austenitic | Must be present | Tensile stress required |
| Intergranular | 450-850C (sensitization range) | Not required | Time at temperature |
| Galvanic | Accelerates all electrochemistry | Increases conductivity | Electrolyte presence |
| Uniform | Higher = faster | Minimal | Strong acid concentration |
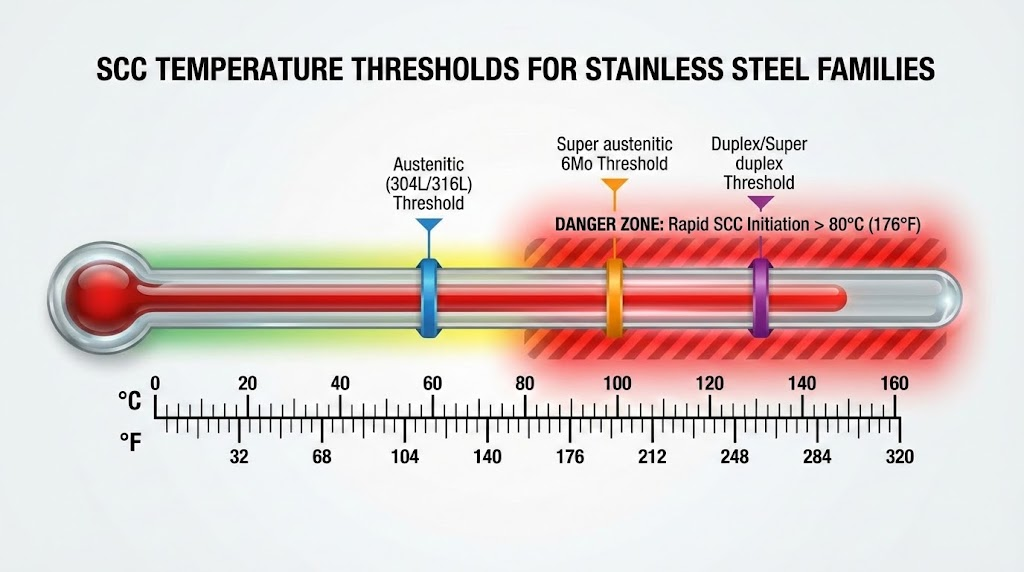
SCC thresholds vary by stainless steel family. For different types of stainless steel and grades, temperature resistance to SCC differs significantly.
Austenitic grades (304L/316L): SCC becomes a risk above 60C when immersed in chloride solutions. However, SCC can occur at 50C or lower in evaporative conditions where chlorides concentrate.
Super austenitic 6Mo grades: Threshold temperature rises to approximately 100C (212F).
Duplex and super duplex grades: Threshold reaches approximately 130C (266F), making these grades preferred for high-temperature chloride environments.
Above 80C, SCC initiation can be rapid. Brief temperature excursions above this threshold are as dangerous as sustained high-temperature operation.
Chloride ions are the primary driver for pitting, crevice corrosion, and SCC.
For 304 stainless steel, pitting can initiate at chloride concentrations as low as 25 ppm. Seawater contains approximately 19,400 ppm chloride, requiring PREN values above 32 for reliable resistance.
Avoid using stainless steel with hydrochloric acid entirely. This acid donates chloride ions directly to the solution, bypassing normal concentration thresholds.
Different acids attack stainless steel through different mechanisms.
| Acid | Safe Range (316L) | Dangerous Range | Recommended Action |
|---|---|---|---|
| Sulfuric | <15% or >85% | 15-85% | Specify higher alloys or non-metallic |
| Phosphoric | Most concentrations | Very high temps | 316/317 adequate for most uses |
| Hydrochloric | None | All concentrations | Use Hastelloy or titanium |
For sulfuric acid, 316L stainless steel provides acceptable corrosion resistance at room temperature only when concentration is below 15% or above 85%. The 15-85% mid-range actively attacks stainless steel.
Intergranular corrosion results from sensitization, chromium carbide precipitation at grain boundaries that depletes nearby chromium. This occurs in the 450-850C (842-1562F) temperature range.
Welding is the primary sensitization risk. The heat-affected zone reaches sensitization temperatures during fabrication. Even low-carbon L grades can sensitize during extended service exposure in this temperature range.
Grade selection for corrosion resistance centers on the Pitting Resistance Equivalent Number (PREN). This calculated value predicts chloride corrosion resistance.
PREN = %Cr + 3.3 x %Mo + 16 x %N
Higher PREN values indicate better resistance to pitting and crevice corrosion. Key thresholds to remember:
PREN comparisons work within the same stainless steel family. Comparing austenitic PREN to duplex PREN directly is not valid because the alloy families have different base corrosion resistance characteristics.
| Grade | PREN Range | Best Applications | Limitations |
|---|---|---|---|
| 304/304L | 17.5-20.8 | General use, mild environments | Avoid high chlorides, seawater |
| 316/316L | 23.1-28.5 | Marine-adjacent, moderate chlorides | Not for warm seawater immersion |
| 904L | 32.2-39.9 | Seawater, acidic environments | Higher cost |
| Duplex 2205 | 30.8-38.1 | Seawater, high stress applications | Better SCC resistance than austenitic |
| Super Duplex 2507 | >40 | Aggressive chloride, subsea | Premium price |
For marine-adjacent applications, 316L is often over-specified for low-temperature uses and under-specified for warm seawater. If your application involves submerged seawater above 30C, specify grades with PREN above 32, such as super duplex or super austenitic alloys.
Prevention strategies must match the specific corrosion mechanism. A single approach cannot address all six types.
When fabricating stainless steel castings, understanding how to weld stainless steel properly minimizes sensitization risk.
Industry standards provide objective methods for verifying corrosion resistance. Request appropriate testing based on your corrosion risks.
| Standard | Test Type | Purpose |
|---|---|---|
| ASTM G48 | Ferric chloride exposure | Pitting and crevice corrosion resistance |
| ASTM G150 | Electrochemical method | Critical pitting temperature (CPT) determination |
| ASTM A262 | Various acid exposures | Intergranular corrosion susceptibility |
| ASTM A967/A380 | Chemical or electrochemical | Passivation verification |
| ASTM B117 | Salt spray chamber | Accelerated corrosion screening |
Salt spray testing per ASTM A967 and A380 requires properly passivated stainless parts to resist corrosion for just 2 hours. This is a minimum threshold, not a performance guarantee.
Selecting the right stainless steel grade and specifying proper heat treatment and surface finishing requires matching your specific operating conditions to material capabilities.
Ready to discuss your corrosion-resistant stainless steel casting requirements? Contact KT Metal Castings with your operating conditions, including temperature range, chloride exposure, and pH levels, for grade recommendations tailored to your application environment.

