Rust costs the world $2.5 trillion every year. That staggering figure represents 3.4% of the global GDP, and it’s all because of a simple chemical reaction between iron and its environment. If you work with metal parts or plan to use custom metal components, understanding what rust is and why it forms will help you avoid expensive problems down the road.
By the end of this guide, you’ll know exactly what causes that reddish-brown coating, which metals are susceptible, and how to prevent it from damaging your projects.

Rust is iron oxide, a reddish-brown compound that forms when iron reacts with oxygen and water. The chemical formula is Fe2O3, and it’s the result of a natural process called oxidation.
Think of it like an apple turning brown after you cut it open. When the apple’s flesh is exposed to air, it reacts with oxygen and changes color. Iron does something similar. When exposed to air and moisture, it forms a completely new substance, which is that familiar reddish-brown coating we call rust.
The color you see isn’t just surface staining. It’s the actual chemical compound itself. This matters because rust isn’t simply sitting on top of the metal. It has replaced some of the original iron.
Here’s something most people don’t realize: only iron and iron-based metals rust. When you see copper turn green or aluminum turn dull gray, that’s not rust. Those are different types of corrosion.
| Metal | What Happens | Appearance | Protects Metal? |
|---|---|---|---|
| Iron/Steel | Rust (iron oxide) | Reddish-brown, flaky | No |
| Copper | Patina (copper carbonate) | Green | Yes |
| Aluminum | Aluminum oxide | Dull gray/white | Yes |
| Brass | Tarnish | Brownish | Yes |
The key difference is what happens next. When aluminum oxidizes, it forms a thin, hard layer that actually shields the metal underneath from further damage. Copper’s famous green patina does the same thing. These protective layers are why copper pipes can last 50 to 70 years and aluminum doesn’t crumble away.
Rust behaves differently. It’s porous and flaky, so it doesn’t seal the surface. Instead, it keeps exposing fresh iron underneath, allowing the rusting process to continue until the metal is completely consumed. That’s why I recommend treating rust as a serious concern rather than just a cosmetic issue.
Rust needs three specific ingredients to form. Remove any one of them, and rusting won’t occur to any significant extent.
All three must be present. A steel beam stored in a completely dry environment will resist rusting for years. The same beam left outdoors in humid conditions can start showing rust within hours.
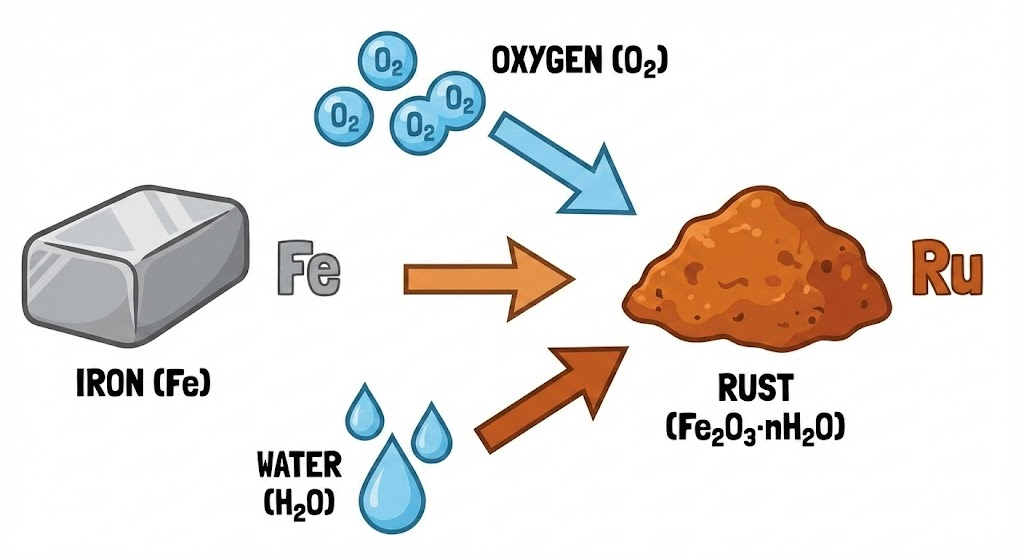
Water is the critical catalyst in the rusting process. It acts as a pathway that allows oxygen to reach the iron and complete the chemical reaction.
Without moisture, the reaction happens so slowly it’s practically invisible. This explains why desert climates preserve metal objects for decades while coastal environments destroy them in years. It also explains why properly storing metal parts in dry conditions is one of the simplest and most effective rust prevention strategies.
Certain conditions accelerate rust formation dramatically:
Under the right conditions, exposed iron can begin rusting within a few hours. That’s why I always recommend addressing rust prevention before a problem develops rather than trying to fix damage after it appears.
Understanding which metals are vulnerable to rust is essential for material selection. This knowledge can save you significant money and maintenance headaches.
These metals contain iron and will rust when exposed to moisture and oxygen:
If you’re working with custom iron castings or custom steel castings, discussing rust protection with your foundry during the design phase is far more effective than applying treatments after production.
These metals don’t rust, though they may corrode in different ways:
For applications where corrosion resistance is critical, aluminum or stainless steel often justify their higher cost through reduced maintenance and longer service life.
Stainless steel deserves special mention because it contains iron but typically doesn’t rust. The secret is chromium content, at least 10.5%.
Chromium protects the steel from rusting because it combines with oxygen before iron does, forming a thin, invisible chromium oxide layer on the surface. This layer is self-healing. If you scratch stainless steel, a new protective layer forms almost immediately.
However, stainless steel can still rust under certain conditions. Deep scratches that deplete the chromium layer, exposure to chlorides, or choosing the wrong grade for your environment can all lead to rust formation. That’s why understanding how long stainless steel takes to rust and whether stainless steel can rust matters for proper material selection.
Prevention is always more effective than treatment. Here are the most common methods for protecting iron and steel from rust.
| Method | How It Works | Best For |
|---|---|---|
| Galvanization | Zinc coating sacrifices itself to protect the iron underneath | Steel structures, outdoor equipment |
| Powder coating | Creates a durable protective barrier | Industrial parts, equipment housings |
| Paint/coatings | Physical barrier blocks moisture and oxygen | General-purpose protection |
| Oil/grease | Moisture barrier that displaces water | Tools, machinery, cast iron cookware |
| Dry storage | Removes moisture from the equation entirely | Storage, maintenance practices |
Galvanized coatings are particularly impressive. A properly applied zinc coating can protect steel for over 100 years by acting as a sacrificial anode. The zinc corrodes instead of the steel underneath, slowly depleting over decades while keeping the base metal intact.
For new projects, material selection often matters more than after-the-fact treatment:
When working with a foundry on custom parts, bring up the operating environment early in the conversation. Understanding whether the part will face humidity, salt exposure, or temperature cycling helps your supplier recommend appropriate materials and finishes.
Now you understand that rust forms when iron, oxygen, and moisture combine, and that it’s a preventable problem rather than an inevitable fate for metal parts. When planning your next project involving metal components, consider the environment those parts will face and discuss material options with your supplier before production begins.
Ready to explore corrosion-resistant options for your metal casting project? Contact our team to discuss which materials and coatings best suit your application.

